Diamine-Functionalized Metal-Organic Framework: Exceptionally High CO2 Capacities from Ambient Air and Flue Gas, Ultrafast CO2 Uptake Rate, and Adsorption Mechanism
슈퍼관리자
2021-05-21
Diamine-Functionalized Metal-Organic Framework: Exceptionally High CO2 Capacities from Ambient Air and Flue Gas, Ultrafast CO2 Uptake Rate, and Adsorption Mechanism
-
Authors :
W. R. Lee, S. Y. Hwang, D. W. Ryu, K. S. Lim, S. S. Han, D. Moon, J. Choi, and C. S. Hong
-
Journal :
Energy & Environmental Science
-
Vol :
7
-
Page :
744-751
-
Year :
2014
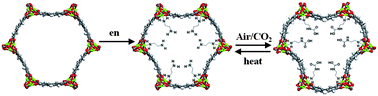
Abstract
he adsorbed CO2 amount of 1-en is 4.57 mmol g−1 (16.7 wt%) at 25 °C and 1 bar and decreases to 3.00 mmol g−1 (11.7 wt%) at 150 °C. Noticeably, 1-en shows a significant CO2 uptake (3.62 mmol g−1, 13.7 wt%) at 0.15 bar, which is comparable to the CO2 partial pressure of a post-combustion flue gas. The CO2 capacity of 1-en at 0.39 mbar, close to atmospheric CO2 concentration, is 2.83 mmol g−1 (11.1 wt%), which marks the highest amount among MOFs. The isosteric heat of adsorption (−Qst) of 1-en in CO2 capture corresponds to 49–51 kJ mol−1, which is supported by DFT calculations (−52.8 kJ mol−1). These results suggest that the adsorption of CO2 onto the free amines of en leads to the formation of a carbamic acid. Adsorption–desorption cyclings of CO2 at the real dilute concentrations of air and flue gas are established with almost retaining CO2 capacities, which could provide superior potential for practical application in CO2 capture. The adsorption rate of CO2 in 1-en exceeds that in some other tested porous materials. The recyclability in CO2 uptake for 1-en is maintained even after exposure to humidity.